tahrir Ghiath Majeed
Assistant Professor Shaza Abdul Amir Jawad
College of Education for Pure Sciences / Department of Chemistry
Azomethine compounds are among the most versatile organic molecules due to their structural flexibility and ability to form stable complexes with transition metals. These complexes exhibit unique physical and chemical properties, making them suitable for various applications, especially in catalysis, analytical chemistry, and optical materials. This study highlights the redox and optical behavior of azomethine compounds and their complexes, along with their scientific significance and potential uses (1).
Figure1.shows application of Schiff base complexes
Azomethines readily coordinate with transition metal ions such as Cu²⁺, Ni²⁺, Pd²⁺, Co²⁺, and Fe³⁺ to form stable complexes with diverse geometries including square planar, tetrahedral, and octahedral. The nature of the ligand and the metal ion plays a significant role in determining the final structure and reactivity (و).
Optical and Spectral Properties:
Azomethine complexes exhibit distinctive optical behaviors, often studied using UV-Vis spectroscopy. These include d-d transitions and ligand-to-metal charge transfer (LMCT) transitions, which impart vivid coloration and, in some cases, fluorescence (3) ,(4)
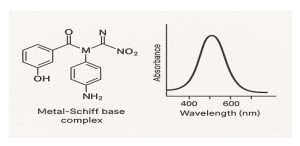
Figur2.Adigital visual representation illustrating the optical changes associated with Schiff base complexes
Modifying the ligand structure allows for fine-tuning of absorption wavelengths and intensities (5) . Certain lanthanide and actinide complexes also exhibit strong fluorescence, opening avenues for photonic applications (6).

Figur3.A schematic diagram illustrating the mechanism of color change in Schiff base complexes due to electronic transition such as d-d transition and metal-to-ligand charge transfer (MLCT)
These transition influence light absorption, resulting in observable optical properties(7). https://youtu.be/4WJeLKKEPLk?si=IfkJhFqNWuy_SHZd.
Reference
1. Cozzi, P. G. (2004). “Metal–Schiff base complexes in catalysis: Recent developments.” Chemical Society Reviews, 33(7), 410–421. https://doi.org/10.1039/B307853C
2. Singh, K., Barwa, M. S., & Tyagi, P. (2006). “Synthesis, characterization and biological studies of Co(II), Ni(II) and Cu(II) complexes with bidentate Schiff bases.” European Journal of Medicinal Chemistry, 41(1), 147–153.
3. Elmali, A. et al. (2000). “Spectral and redox properties of some transition metal-Schiff base complexes.” Transition Metal Chemistry, 25, 491–494.
4. Gupta, K. C., & Sutar, A. K. (2008). “Catalytic activities of Schiff base transition metal complexes.” Coordination Chemistry Reviews, 252(12), 1420–1450.
5. Yadav, R. P., & Srivastava, A. K. (2014).Colorimetric sensing of metal ions using Schiff base complexes.
Sensors and Actuators B: Chemical, 195, 260-265.
https://doi.org/10.1016/j.snb.2014.01.058
6. Wang, H. et al. (2013).
A novel colorimetric sensor for metal ion recognition based on Schiff base derivatives.
Talanta, 107, 361–366.
https://doi.org/10.1016/j.talanta.2012.12.036
7. Akkurt, M. et al. (2018).
Synthesis and colorimetric properties of metal complexes derived from Schiff bases.
Journal of Molecular Structure, 1153, 39–46.
https://doi.org/10.1016/j.molstruc.2017.10.056.





























































